Research topics
Multi-scale Molecular Simulation of Polymer Degradation
The increasing international focus on global environmental issues, including the Sustainable Development Goals (SDGs), has highlighted the importance of “long-term use of materials.” In particular, the use and dispose of polymer materials, which are closely linked to the issue of plastic waste. Therefore, understanding the “degradation phenomena” that occur due to exposure to UV and heat in the actual environmental conditions in which these materials are used is of paramount importance.
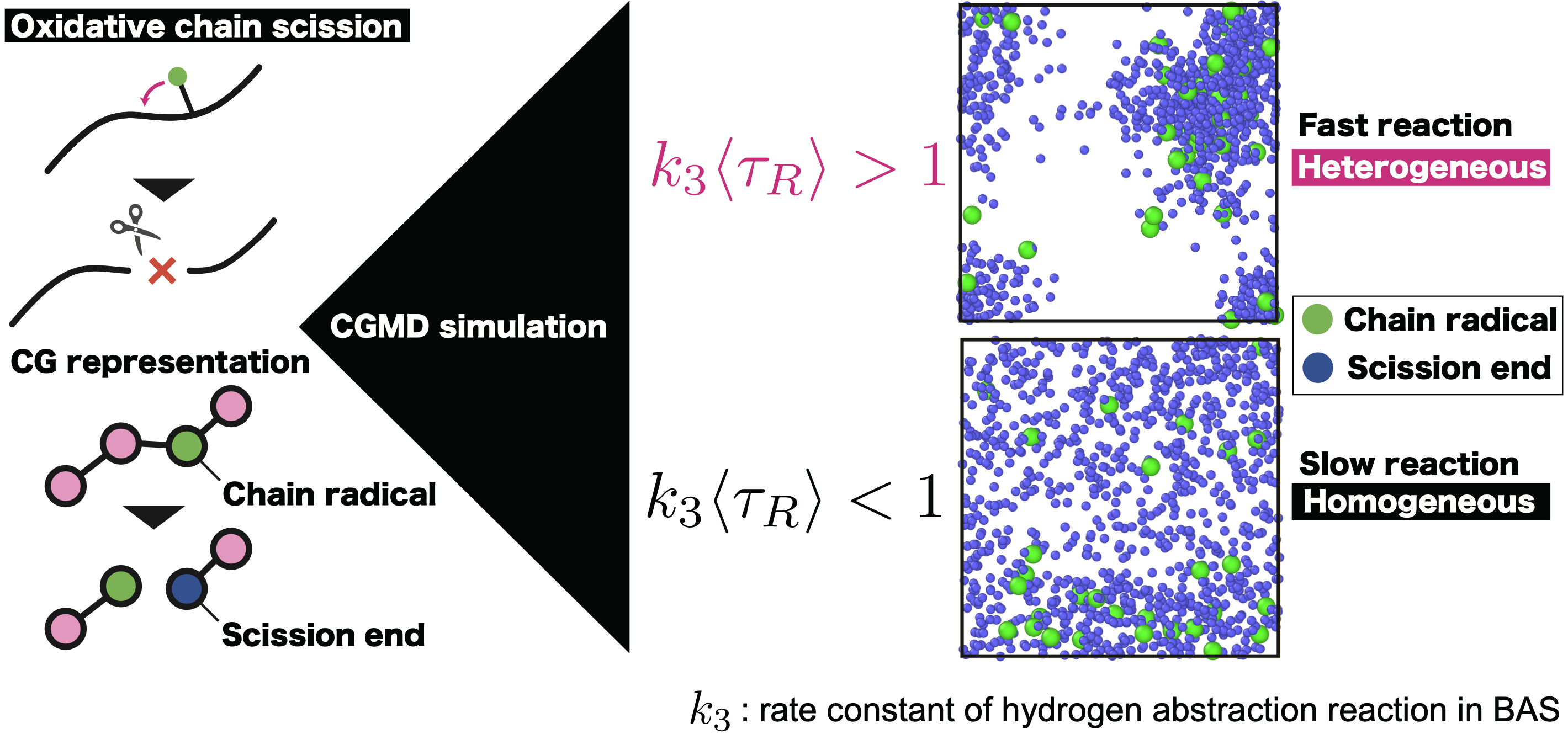
Most of the gradual degradation of materials over long-term usage is caused by “oxidative degradation,” which involves the reaction with oxygen in the air and radicals. Radicals are highly reactive and can be considered a sort of “source of aging”. It was visualized in experiments in the 2000s that the degradation of polymers progresses in a manner similar to the spread of an infectious disease, where radicals, the source of degradation, propagate the reaction. This phenomenon has been represented computationally, and molecular simulation methods that are consistent with the actual mechanisms of oxidatve aging have been established.
Research to date has shown that the heterogeneity of oxidative degradation in micro/meso scales arises from the competition between the timescales of radical reactions and the relaxation time of polymers. Recent research efforts are focused on extending these findings to larger, macroscopic scales. This progression of research from micro to macro scales is essential for developing a comprehensive understanding of polymer degradation, which can inform strategies for material design, recycling, and sustainability aligned with global environmental goals.
References
- T. Ishida*, Y. Doi, T. Uneyama, Y. Masubuchi, Modeling for heterogeneous oxidative aging of polymers using coarse-grained molecular dynamics, Macromolecules, 56(21), 8474-8483, 2023.
Polymer Decomposition Simulation Towards Acceleration of Chemical Recycling
Chemical recycling is a recycling method that has garnered significant attention for its potential contribution to achieving carbon neutrality. This method involves the chemical decomposition of plastics, converting them back into clean chemical raw materials, thereby enabling the complete recycling of resin materials. Although the resource recovery efficiency of chemical recycling technologies still has room for improvement, it is suggested that optimization of the decomposition process design, through integration with computational science, could lead to a reduction in CO2 emissions by more than 100 million tons annually. Our aim is to accelerate chemical recycling with the aid of computer simulations. Currently, we have developed a method for representing pyrolysis, a robust and economical process often employed, using coarse-grained molecular simulations in computational models.

References
- T. Ishida*, Y. Doi, T. Uneyama, Y. Masubuchi, Modeling for heterogeneous oxidative aging of polymers using coarse-grained molecular dynamics, Macromolecules, 56(21), 8474-8483, 2023.
Characterization of Polymer Degradation in Collaboration with Advanced Analytical Chemistry
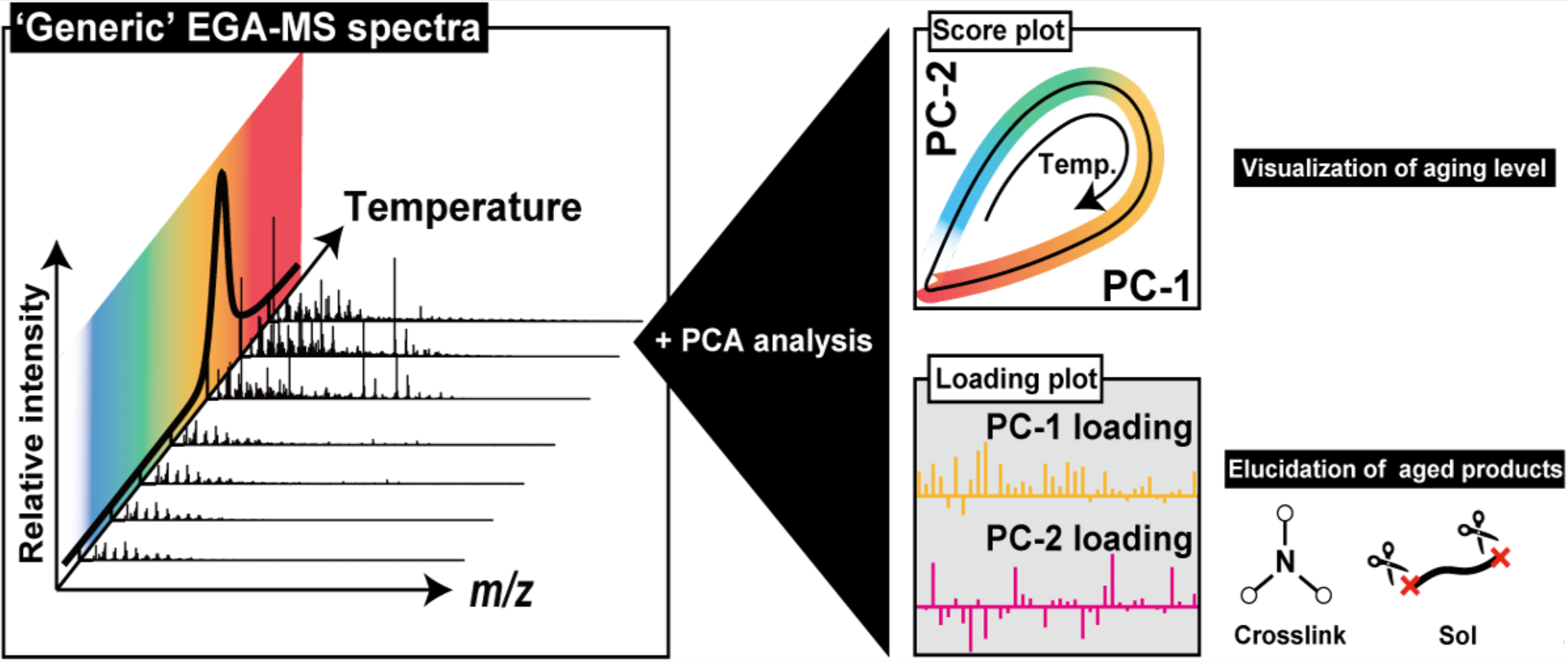
To characterize the polymer aging, it is essential to collect and interpret evidence from multiple perspectives by combining various analytical chemistry methods. Analytical instruments are rapidly evolving, and the detailed analytical data they produce can now be considered as big data. Additionally, as polymers degrade, their chemical and higher-order structures become much more complex, making it challenging to interpret the analytical data and identify characteristic signals of the aging mechanisms. Systematically varying aging time and employing informatics (data science) methods to analyze collected mass spectrometry and optical spectroscopy data can facilitate a deeper understanding of these mechanisms. Analytical methods that have proven effective in such analysis include Fourier Transform Infrared Spectroscopy (FTIR), Evolved Gas Analysis-Mass Spectrometry (EGA-MS), and Pyrolysis Gas Chromatography-Mass Spectrometry (PyGC-MS).
References
- T. Ishida*, R. Kitagaki, Y. Elakneswaran, J. Mizukado, H. Shinzawa, H. Sato, H. Hagihara, R. Watanabe, Network Degradation Assessed by Evolved Gas Analysis-Mass Spectrometry Combined with Principal Component Analysis (EGA-MS-PCA): A Case of Thermo-oxidized Epoxy/Amine Network, Macromolecules, 56 (3), 883–891, 2023.
- T. Ishida*, R. Watanabe, H. Hagihara, J. Mizukado, R. Kitagaki, Y. Elakneswaran, In-situ Infrared Cure Monitoring Coupling with Two-Trace Two- Dimensional (2T2D) Correlation Analysis can Elucidate Property- Enhancement Mechanisms due to Nanofiller Incorporation on Thermosetting Polymer, Polymer Testing, 112, 107587, 2022.
- T. Ishida*, R. Kitagaki, R. Watanabe, H. Hagihara, Y. Elakneswaran, H. Shinzawa, A study of molecular architectural dynamics of crosslinked urethane during photo-aging by two-dimensional infrared correlation spectroscopy, Polymer Degradation and Stability, 179, 109242, 2020.
Kinetic Modeling of Radical Decomposition Reactions
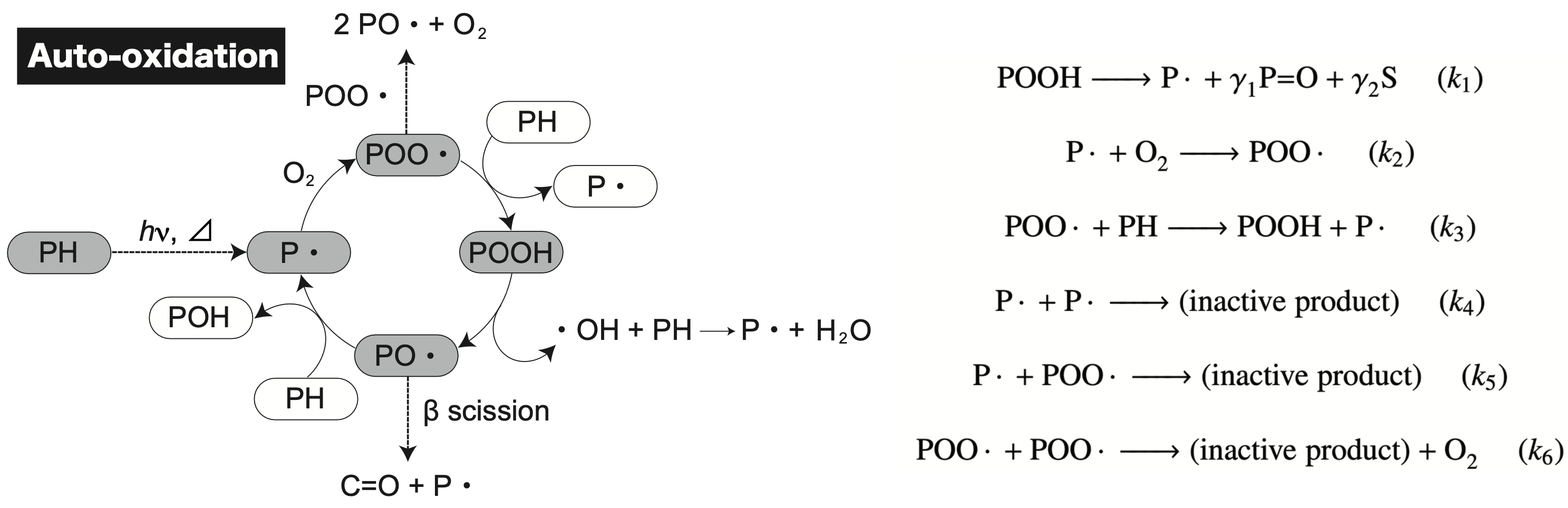
I am engaged in constructing kinetic models of radical reactions for several systems. Until now, my focus has primarily been on developing the kinetics of oxidative degradation. This involves accounting for elementary processes such as scission and cross-linking that occur within the autoxidation mechanism. By quantifying these molecular chain transformations, it is possible to connect changes in molecular weight distribution and physical properties associated with degradation. This approach allows for a more comprehensive understanding of the aging process by linking the underlying chemical transformations to the observable changes in the material’s properties.
References
- T. Ishida*, E. Richaud, M. Gervais, A. Gaudy, R. Kitagaki, H. Hagihara, Y. Elakneswaran, Thermal aging of acrylic-urethane network: Kinetic modeling and end-of-life criteria combined with mechanical properties, Prog. Org. Coatings, 163, 106654, 2022.
Theory and Simulation of Diffusion of Lightweight Molecules in Cementitious Materials
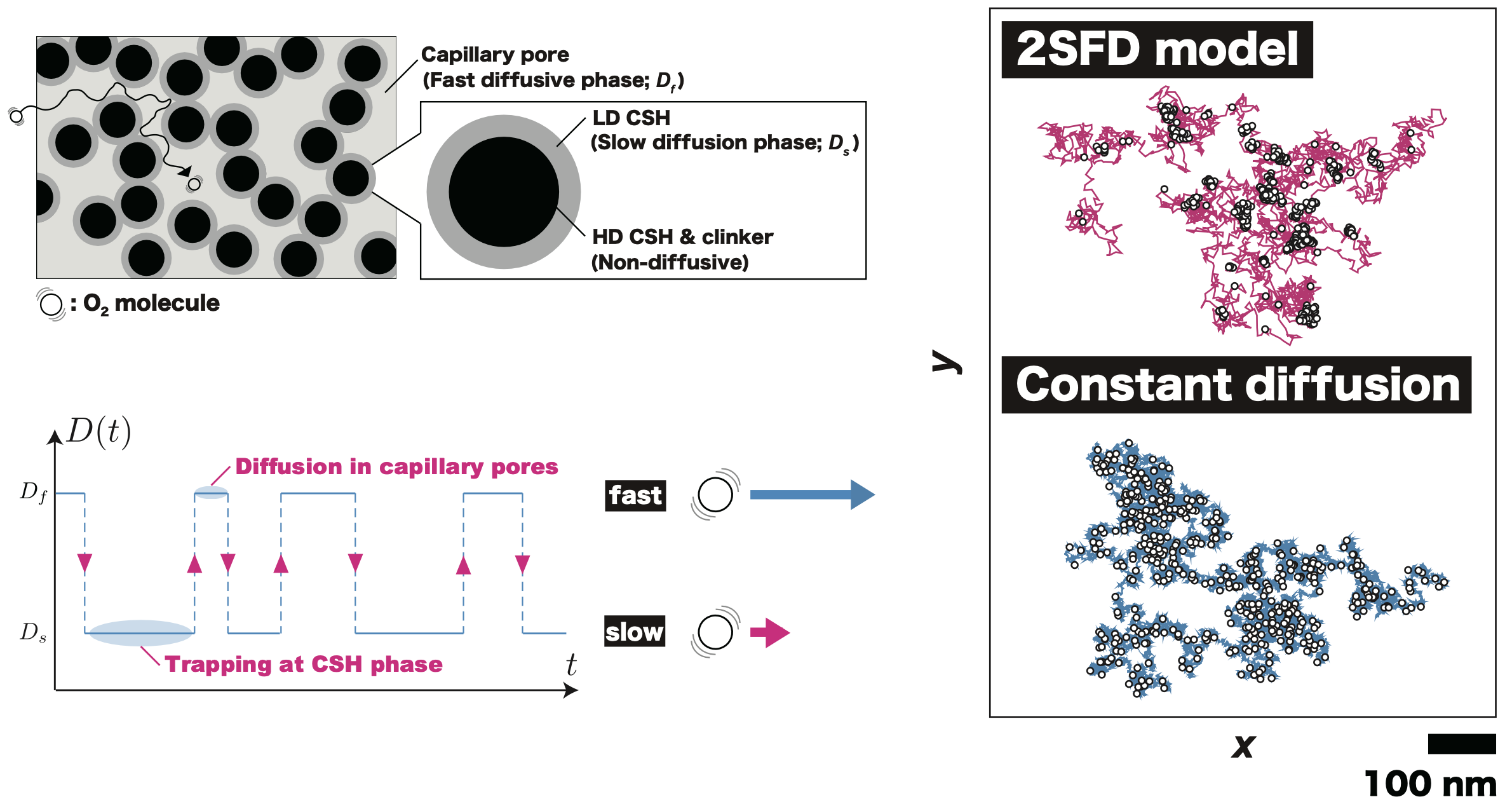
This topic stems from my personal interest in building materials, kind a hobby. The diffusion of low molecular weight species in cementitious materials is a crucial phenomenon deeply related to the lifespan of reinforced concrete structures, integrating to daily life. The internal steel bar is protected by being encased in pure concrete. However, the structural integrity is compromised by the infiltration of low molecular weight species (such as carbon dioxide, water, chloride ions, sulfate ions) that can cause or exacerbate rusting of the reinforcement.
I have successfully applied the framework of “fluctuating diffusivity”, a concept advanced in the field of statistical physics, to describe the diffusion of low molecular weight species in cementitious materials. This application revealed that in the short-time domain, there exists a region where the distribution of diffusion displacements shows a heavy-tailed, non-Gaussian behavior, differing from predictions based on traditional effective diffusion equations. The persistence of this non-Gaussianity over certain time periods could have implications for the reliability assessment of structures. This ongoing challenge in the field of cement and concrete research involves describing this diffusion phenomenon accurately, which is central to understanding and predicting the long-term behavior of concrete structures.
References
- F. Nakai, T. Ishida*, Gas Diffusion in Cement Pastes: An Analysis using a Fluctuating Diffusivity Model, Construction and Building Materials, 407(1), 133411, 2023.







